 
Individuals aged five years and older can receive a booster dose of a mRNA COVID-19 vaccine.  
Booster doses help keep you protected from severe outcomes from the virus. Protection after a primary series may decrease over time, especially against new variants. Who is eligible for a COVID-19 vaccine booster dose?īooster doses are doses of a COVID-19 vaccine received after the primary series. Please note: the AstraZeneca, Johnson & Johnson (Janssen), and Medicago Covifenz vaccines are not available for administration in Ontario.įor more information about the COVID-19 vaccine primary series, please refer to the Ministry of Health’s COVID-19 Vaccine Guidance and COVID-19 Vaccines webpage. Contact Public Health via email at to receive information on how to request the Novavax vaccine. Please note that Public Health vaccine clinics do not administer this product. The Novavax vaccine is a two-dose recombinant protein subunit vaccine authorized for individuals 12 years of age and older. Those who are not able to or do not wish to receive an mRNA COVID-19 vaccine series or booster can receive the Novavax COVID-19 vaccine via their primary care provider or local pharmacy. This recommendation is based on evidence that suggests longer intervals between first and second doses of COVID-19 vaccines result in a more robust and durable immune response and higher vaccine effectiveness. Most individuals are recommended to receive a second dose at an interval of eight weeks (56 days) after their first dose to complete the primary series, or as directed by their health care provider. Individuals over the age of 5 may receive any combination of COVID-19 mRNA vaccines to complete their primary series. It is recommended that infants and children under 5 years of age receive the same vaccine product for all primary series doses, using the vaccine that is correct for their age at the time of administration. The National Advisory Committee on Immunization (NACI) recommends mRNA vaccines (Pfizer or Moderna) for both primary series and booster dose(s), with the preference of Pfizer for individuals aged 5 to 29. Most people need two doses of an mRNA vaccine ( Pfizer, Moderna) or Novavax (for those over 12 years of age) to complete their primary series.Ĭhildren under the age of 5 and individuals who are immunocompromised may need a three to four dose primary series. COVID-19 Vaccine eligibility Who is eligible for the COVID-19 vaccine first and second dose (primary series)?Īnyone who is six months and older is eligible to receive a COVID-19 vaccineĪ primary series is the initial number of doses of a COVID-19 vaccine that a person needs to develop a strong initial immune response. All residents are encouraged to get the number of doses they are recommended to receive as soon as they can to ensure maximum protection against COVID-19. The COVID-19 vaccine is free for everyone in Ontario. On the Provincial COVID-19 Vaccination Portal You can walk-in at select clinics or book an appointment through the Provincial COVID-19 Vaccination Portal, or by calling the Provincial Vaccine Contact Centre at 1-83 (TTY for people who are deaf, hearing-impaired or speech-impaired: 1-86). Please contact your health care provider to discuss optimal timing for your next COVID-19 vaccination dose Individuals at high risk for severe COVID-19 illness can receive a booster dose after an interval of six months, and are recommended to book an appointment for a spring COVID-19 vaccine booster. Recommendations for individuals who are not high-risk and have already received a booster since September 1, 2022, will be available closer to Fall 2023. Sunnyside Wellness Centre Toggle SectionĪnyone six months and older is eligible to receive a COVID-19 vaccine.Īnyone five years of age or older who has not yet received a booster dose since September 1, 2022, is encouraged to receive a booster dose at a recommended interval of six months after the completion of their primary vaccine series, last booster dose, or confirmed COVID-19 infection. 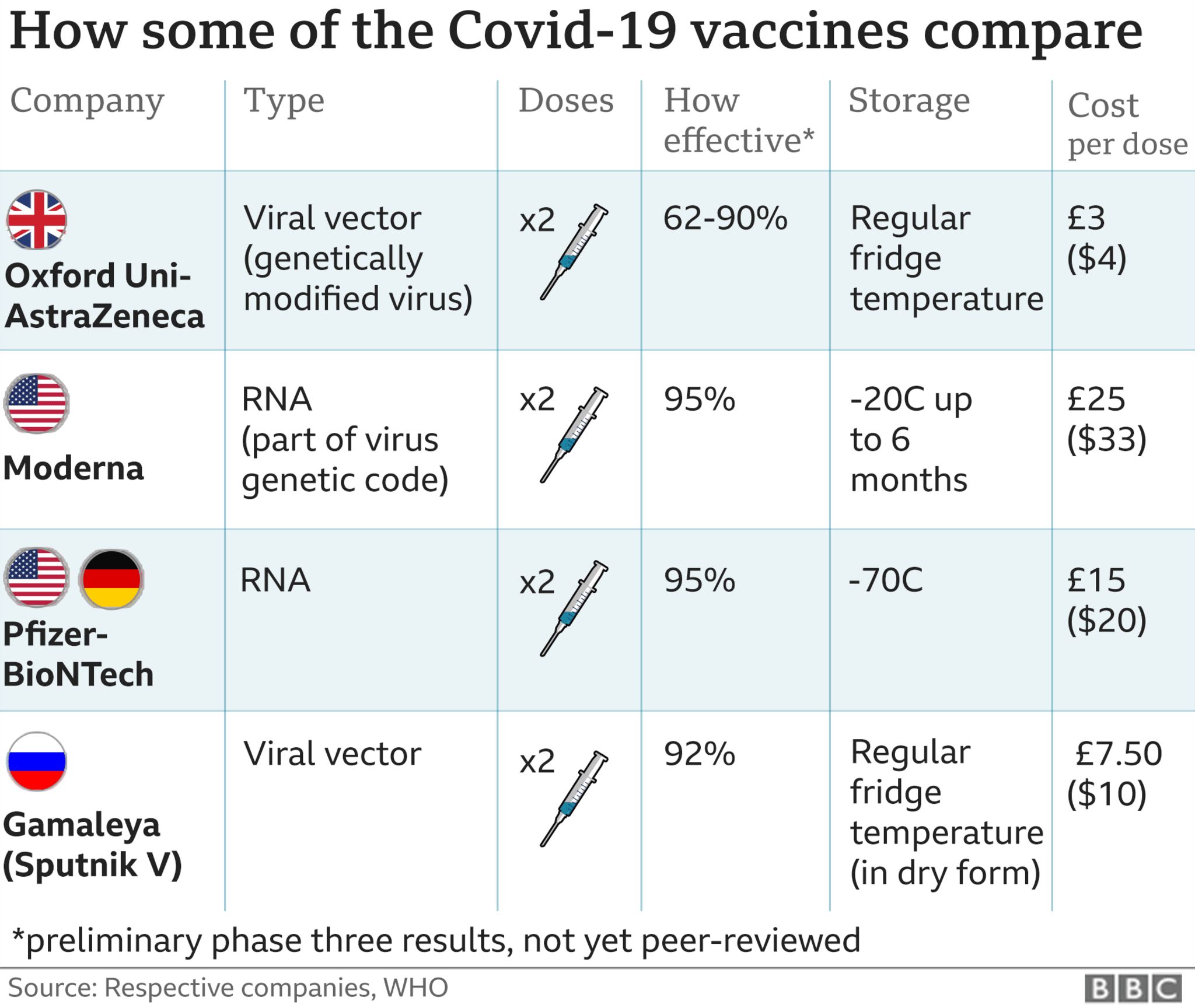
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
